11 results
Validation of antibiotic stewardship metrics for genitourinary infection management in Veterans Affairs outpatient settings
- Jordan Braunfeld, Matthew Samore, Jacob Crook, McKenna Nevers, Kelly Echevarria, Ben Brintz, Matthew Goetz, Karl Madaras-Kelly
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s35
-
- Article
-
- You have access Access
- Open access
- Export citation
-
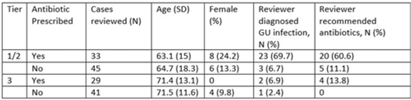
Background: Diagnosis and management of suspected urinary tract infection (UTI) in outpatient settings has been shown to be suboptimal. We previously developed a set of stewardship metrics for UTIs based on electronic health record (EHR) data (Antimicrobial Stewardship & Healthcare Epidemiology 2022;2 suppl 1:S5–S6. doi:10.1017/ash.2022). A tier-based approach was used to more fully capture antibiotic use associated with genitourinary (GU) symptoms and diagnoses. Herein we report a preliminary analysis of validity and reliability of these metrics based on chart abstraction. Methods: The study cohort consisted of patients who visited Veterans Affairs emergency departments or primary care clinics between 2015 and 2022 and who had a GU diagnosis based on International Classification of Disease, Tenth Revision (ICD-10) codes, divided into 3 categories: tier 1 (antibiotics always indicated), tier 2 (antibiotics sometimes indicated), and tier 3 (antibiotics not indicated). Visits related to urological procedures, nontarget settings, or concomitant non-GU infections were excluded. Cases were randomly sampled for manual review from within 8 strata based on tier, use of antibiotics, and visit type. An infectious disease physician and pharmacist abstracted charts using a standardized data-collection instrument. Clinical judgments regarding diagnosis and treatment were recorded on a Likert scale without knowledge of how the patient was managed. The intraclass correlation coefficient (ICC) was used to estimate interrater reliability. Results: To date, 148 cases have been reviewed (50 by both reviewers). Mean (SD) age was 67.5 (15.3) years and 12.2% were female. In a majority of tier 1 and 2 visits in which antibiotics were given, the reviewers found evidence for GU infection (69.7%) and favored prescribing of antibiotics (60.6%) (Table). In contrast, most patients in the tier 3 category who received antibiotics were judged to have noninfectious conditions (eg, benign prostatic hypertrophy) and to not require antibiotics. In the subset of records examined by both reviewers, the interrater reliability of judgments of whether antibiotics were warranted was good (ICC = .704). Conclusions: This preliminary validation provides support for a tier-based approach for stewardship metrics for GU conditions that relies upon electronic data to identify patients for whom antibiotics are generally not indicated.
Disclosures: None
Electronic surveillance criteria for non–ventilator-associated hospital-acquired pneumonia: Assessment of reliability and validity
- Sarah E. Stern, Matthew A. Christensen, McKenna R. Nevers, Jian Ying, Caroline McKenna, Shannon Munro, Chanu Rhee, Matthew H. Samore, Michael Klompas, Barbara E. Jones
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 11 / November 2023
- Published online by Cambridge University Press:
- 15 March 2023, pp. 1769-1775
- Print publication:
- November 2023
-
- Article
- Export citation
-
Objective:
Surveillance of non–ventilator-associated hospital-acquired pneumonia (NV-HAP) is complicated by subjectivity and variability in diagnosing pneumonia. We compared a fully automatable surveillance definition using routine electronic health record data to manual determinations of NV-HAP according to surveillance criteria and clinical diagnoses.
Methods:We retrospectively applied an electronic surveillance definition for NV-HAP to all adults admitted to Veterans’ Affairs (VA) hospitals from January 1, 2015, to November 30, 2020. We randomly selected 250 hospitalizations meeting NV-HAP surveillance criteria for independent review by 2 clinicians and calculated the percent of hospitalizations with (1) clinical deterioration, (2) CDC National Healthcare Safety Network (CDC-NHSN) criteria, (3) NV-HAP according to a reviewer, (4) NV-HAP according to a treating clinician, (5) pneumonia diagnosis in discharge summary; and (6) discharge diagnosis codes for HAP. We assessed interrater reliability by calculating simple agreement and the Cohen κ (kappa).
Results:Among 3.1 million hospitalizations, 14,023 met NV-HAP electronic surveillance criteria. Among reviewed cases, 98% had a confirmed clinical deterioration; 67% met CDC-NHSN criteria; 71% had NV-HAP according to a reviewer; 60% had NV-HAP according to a treating clinician; 49% had a discharge summary diagnosis of pneumonia; and 82% had NV-HAP according to any definition according to at least 1 reviewer. Only 8% had diagnosis codes for HAP. Interrater agreement was 75% (κ = 0.50) for CDC-NHSN criteria and 78% (κ = 0.55) for reviewer diagnosis of NV-HAP.
Conclusions:Electronic NV-HAP surveillance criteria correlated moderately with existing manual surveillance criteria. Reviewer variability for all manual assessments was high. Electronic surveillance using clinical data may therefore allow for more consistent and efficient surveillance with similar accuracy compared to manual assessments or diagnosis codes.
Implementation and outcomes of a clinician-directed intervention to improve antibiotic prescribing for acute respiratory tract infections within the Veterans’ Affairs Healthcare System
- Karl J. Madaras-Kelly, Suzette A. Rovelsky, Robert A. McKie, McKenna R. Nevers, Jian Ying, Benjamin A. Haaland, Chad L. Kay, Melissa L. Christopher, Lauri A. Hicks, Mathew H. Samore
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 5 / May 2023
- Published online by Cambridge University Press:
- 15 August 2022, pp. 746-754
- Print publication:
- May 2023
-
- Article
- Export citation
-
Objective:
To determine whether a clinician-directed acute respiratory tract infection (ARI) intervention was associated with improved antibiotic prescribing and patient outcomes across a large US healthcare system.
Design:Multicenter retrospective quasi-experimental analysis of outpatient visits with a diagnosis of uncomplicated ARI over a 7-year period.
Participants:Outpatients with ARI diagnoses: sinusitis, pharyngitis, bronchitis, and unspecified upper respiratory tract infection (URI-NOS). Outpatients with concurrent infection or select comorbid conditions were excluded.
Intervention(s):Audit and feedback with peer comparison of antibiotic prescribing rates and academic detailing of clinicians with frequent ARI visits. Antimicrobial stewards and academic detailing personnel delivered the intervention; facility and clinician participation were voluntary.
Measure(s):We calculated the probability to receive antibiotics for an ARI before and after implementation. Secondary outcomes included probability for a return clinic visits or infection-related hospitalization, before and after implementation. Intervention effects were assessed with logistic generalized estimating equation models. Facility participation was tracked, and results were stratified by quartile of facility intervention intensity.
Results:We reviewed 1,003,509 and 323,023 uncomplicated ARI visits before and after the implementation of the intervention, respectively. The probability to receive antibiotics for ARI decreased after implementation (odds ratio [OR], 0.82; 95% confidence interval [CI], 0.78–0.86). Facilities with the highest quartile of intervention intensity demonstrated larger reductions in antibiotic prescribing (OR, 0.69; 95% CI, 0.59–0.80) compared to nonparticipating facilities (OR, 0.89; 95% CI, 0.73–1.09). Return visits (OR, 1.00; 95% CI, 0.94–1.07) and infection-related hospitalizations (OR, 1.21; 95% CI, 0.92–1.59) were not different before and after implementation within facilities that performed intensive implementation.
Conclusions:Implementation of a nationwide ARI management intervention (ie, audit and feedback with academic detailing) was associated with improved ARI management in an intervention intensity–dependent manner. No impact on ARI-related clinical outcomes was observed.
Using machine learning to predict antibiotic resistance to support optimal empiric treatment of urinary tract infections
- Ben Brintz, McKenna Nevers, Matthew Goetz, Kelly Echevarria, Karl Madaras-Kelly, Matthew Samore
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s69
-
- Article
-
- You have access Access
- Open access
- Export citation
-
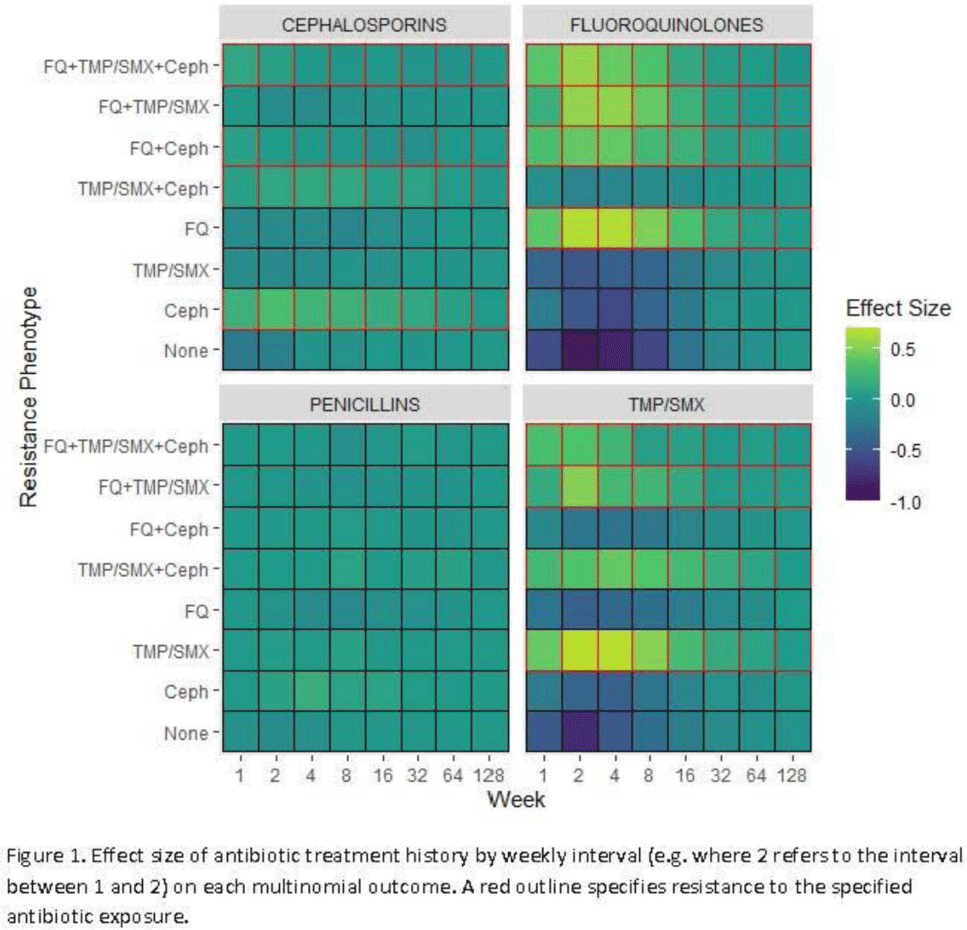
Background: Antibiotic resistance is pervasive in the Veterans’ Affairs (VA) healthcare system, with rates of fluoroquinolone and trimethoprim–sulfamethoxazole (TMP/SMX) resistance approaching 30% in E. coli urinary isolates. The efficacy of antimicrobial treatment is critically dependent on the susceptibility of the infecting pathogen; however, prescription decisions are often made empirically in practice. We analyzed susceptibility profiles of enteric gram-negative rods (Enterobacterales) from clinical urine cultures collected from ambulatory patients receiving care in VA clinics and emergency departments. Our goals were (1) to develop a predictive model to support choice of empiric antibiotics pending results of susceptibility testing and (2) to examine the relationship between past antibiotic exposures and susceptibility profiles to enhance understanding of antibiotic selective pressure. Methods: We obtained 265,076 positive cultures from 157,422 unique patients from 2015 to 2020. We trained random forest multinomial classifiers to estimate the risk of a positive urine culture isolate being resistant to the multinomial outcome: fluoroquinolone, TMP–SMX, cephalosporin, or any combination of these 3 agents. Data sources evaluated for model generation included demographics, comorbidities, trend and seasonal terms, treatment history for multiple antimicrobial treatments summarized using number of prescriptions in weekly intervals, and sample history summarized by number of resistant and susceptible cultures in weekly intervals. Using 5-fold cross validation, we assess the performance of the clinical prediction using the area under the receiver operating characteristic curve (AUC) for each multinomial outcome. In addition to prediction, we modeled the direct effect of treatment on resistance using multinomial group lasso (MGL). This method allows variable selection in variable groupings, such as all variables related to the fluoroquinolone treatment history, which allowed us to assess the effect of a patient’s complete course of treatment on resistance. Results: In cross-validation analysis, our random forest model was best at predicting outcomes with fluoroquinolone resistant phenotypes compared to non–fluoroquinolone-resistant phenotypes (Table 1). From MGL, we found that having a prescription for fluoroquinolone treatment 4–8 weeks prior to a urinalysis was positively associated with fluoroquinolone resistance and negatively associated with fluoroquinolone susceptible phenotypes (Fig. 1). Conclusions: Our results show that a patient’s sample and treatment history are highly predictive of a future resistance. Fluoroquinolone treatment is especially associated with increased risk of fluoroquinolone single- and multidrug resistances. A history of either fluoroquinolone or trimethoprim-sulfamethoxazole (TMP-SMX) treatment is a stronger indicator of a future resistant phenotype than cephalosporin or penicillin.
Funding: None
Disclosures: None

Tier-based antimicrobial stewardship metrics for genitourinary-related antibiotic use in Veterans’ Affairs outpatient settings
- Matthew Samore, Matthew Goetz, McKenna Nevers, Jacob Crook, Suzette Rovelsky, Ben Brintz, Kelly Echevarria, Melinda Neuhauser, Sharon Tsay, Lauri Hicks, Karl Madaras-Kelly
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s5-s6
-
- Article
-
- You have access Access
- Open access
- Export citation
-
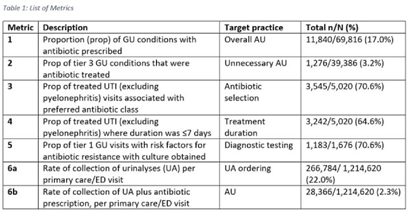
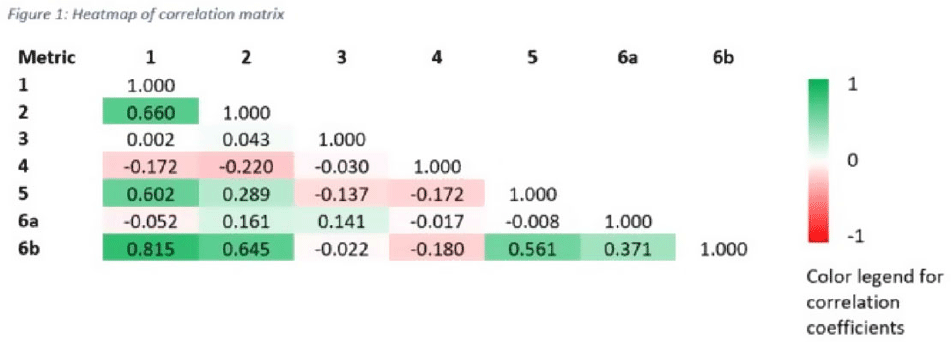
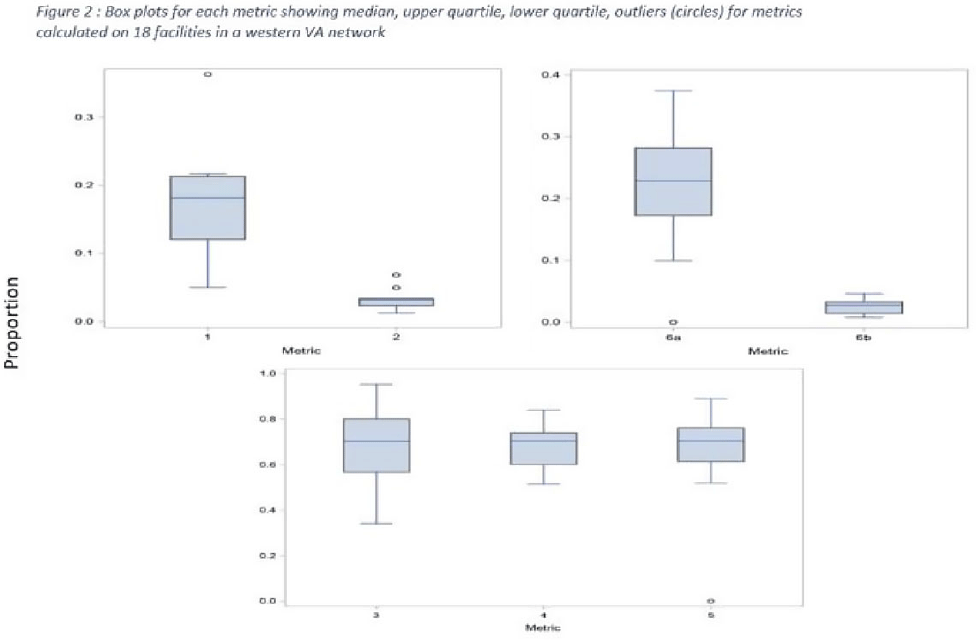
Background: Tracking antibiotic use is a core element of antimicrobial stewardship. We developed a set of metrics based on electronic health record data to support an outpatient stewardship initiative to improve management of urinary tract infections (UTIs) in Veterans’ Affairs (VA) emergency departments (EDs) and primary care clinics. Because UTI diagnostic codes only capture a portion of genitourinary (GU)-related antibiotic use, a tier-based approach was used to evaluate practices. Methods: Metrics were developed to target practices related to antibiotic prescribing and diagnostic testing (Table 1). GU conditions were divided into 3 categories: tier 1, conditions for which antibiotics are usually or always indicated; tier 2, conditions for which antibiotics are sometimes indicated; and tier 3, conditions for which antibiotics are rarely or never indicated (eg, benign prostatic hypertrophy with symptoms). Patients with visits related to urological procedures, nontarget providers, and concomitant non-GU infections were excluded. Descriptive analyses included calculation of the correlation matrix for the 7 metrics and the construction of box plots to display interfacility variability. Results: Metrics were calculated quarterly for 18 VA medical centers, including affiliated clinics, in a western VA network, from July 2018 to June 2020 (Table 1). Tier 3 GU conditions accounted for 1,276 of 11,840 (11%) of GU-related antibiotic use. Metrics 1 and 6b were strongly correlated with each other and were also positively correlated with metrics 2 and 5 (coefficients > 0.5) (Fig. 1). Substantial interfacility variation was observed (Fig. 2). Conclusions: Stewardship metrics for suspected or documented UTIs can identify opportunities for practice improvement. Broadly capturing GU conditions in addition to UTIs may enhance utility for performance feedback. Antibiotic prescribing for tier 3 GU conditions is analogous to unnecessary antibiotic use for acute, uncomplicated bronchitis and upper respiratory tract infections.
Funding: None
Disclosures: None
A Veterans’ Healthcare Administration (VHA) antibiotic stewardship intervention to improve outpatient antibiotic use for acute respiratory infections: A cost-effectiveness analysis
- Minkyoung Yoo, Karl Madaras-Kelly, McKenna Nevers, Katherine E. Fleming-Dutra, Adam L. Hersh, Jian Ying, Ben Haaland, Matthew Samore, Richard E. Nelson
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 10 / October 2022
- Published online by Cambridge University Press:
- 29 September 2021, pp. 1389-1395
- Print publication:
- October 2022
-
- Article
- Export citation
-
Objectives:
The Core Elements of Outpatient Antibiotic Stewardship provides a framework to improve antibiotic use, but cost-effectiveness data on implementation of outpatient antibiotic stewardship interventions are limited. We evaluated the cost-effectiveness of Core Element implementation in the outpatient setting.
Methods:An economic simulation model from the health-system perspective was developed for patients presenting to outpatient settings with uncomplicated acute respiratory tract infections (ARI). Effectiveness was measured as quality-adjusted life years (QALYs). Cost and utility parameters for antibiotic treatment, adverse drug events (ADEs), and healthcare utilization were obtained from the literature. Probabilities for antibiotic treatment and appropriateness, ADEs, hospitalization, and return ARI visits were estimated from 16,712 and 51,275 patient visits in intervention and control sites during the pre- and post-implementation periods, respectively. Data for materials and labor to perform the stewardship activities were used to estimate intervention cost. We performed a one-way and probabilistic sensitivity analysis (PSA) using 1,000,000 second-order Monte Carlo simulations on input parameters.
Results:The proportion of ARI patient-visits with antibiotics prescribed in intervention sites was lower (62% vs 74%) and appropriate treatment higher (51% vs 41%) after implementation, compared to control sites. The estimated intervention cost over a 2-year period was $133,604 (2018 US dollars). The intervention had lower mean costs ($528 vs $565) and similar mean QALYs (0.869 vs 0.868) per patient compared to usual care. In the PSA, the intervention was dominant in 63% of iterations.
Conclusions:Implementation of the CDC Core Elements in the outpatient setting was a cost-effective strategy.
Increased Return Clinic Visits for Adults with Group A Streptococcal Pharyngitis Treated with a Macrolide
- Suzette Rovelsky, Benjamin Pontefract, McKenna Nevers, Adam Hersh, Matthew Samore, Karl Madaras-Kelly
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s295-s296
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: A multicenter audit-and-feedback intervention was conducted to improve management of acute respiratory infections (ARIs) including group A streptococcal (GAS) pharyngitis within 6 VA medical Centers (VAMCs). A relative reduction (24.8%) in azithromycin prescribing after the intervention was observed. Within these facilities during 2015–2018, 2,266 cases of GAS occurred, and susceptibility to erythromycin ranged from 55% to 70%. We evaluated whether prescribing a macrolide for GAS pharyngitis was associated with an increase in outpatient return visits. Methods: A cohort of ambulatory adults treated for GAS pharyngitis (years 2014–2019) at 6 VAMCs was created. Demographic, diagnostic, treatment, and revisit data were extracted from the Corporate Data Warehouse. GAS pharyngitis was defined by an acute pharyngitis diagnostic code combined with a GAS-positive rapid strep test or throat culture ≤3 days of index date. Antibiotic prescriptions were included if filled ≤3 days of index date and were classified as first line (penicillin/amoxicillin), second line (cephalexin/clindamycin), macrolides (azithromycin, clarithromycin, erythromycin), or other (remaining antibiotics). A return visit was defined as a new visit to primary care, urgent care, or the emergency department with a diagnostic code for an ARI ≤30 days from the index visit. Logistic regression was used to adjust for nonantibiotic covariates and to compare treatments. Results are reported as odds ratio (OR ± 95% CI; P value). Results: Of 12,666 patients with a diagnostic code for acute pharyngitis, 2,923 (23.1%) had GAS testing performed. Of those, 582 (19.9%) were GAS-positive and 460 (15.7%) received antibiotics. The mean age was 39.0 years (±SD, 11.7) and 73.7% were male. Antibiotics included penicillins for 363 patients (78.9%), cephalosporins for 21 (4.6%), clindamycin for 32 (7.0%), macrolides for 47 (10.2%), and other for 17 (3.9%). Penicillin allergy was documented in 48 patients (10.5%), and these patients received cephalosporins (18.8%), clindamycin (35.4%), macrolides (41.7%), and other antibiotics (4.2%). Return visits occurred in 47 cases (10.4%). Limited chart review indicated that 6 of 10 macrolide recipients (60.0%) with return visits had recurrence or unresolved symptoms. After adjustment for calendar month and facility, odds of a return visit for treatment with a macrolide relative to penicillins was 2.79 (OR, 1.19; 95% CI, ±6.56; P = .02). The audit-feedback intervention was not associated with ARI-related return visits (OR, 0.53; 95% CI, 0.26–1.06; P = .07). Conclusions: Return visit rates were higher for GAS pharyngitis patients treated with a macrolide than for those treated with penicillins. Macrolides were the most commonly prescribed non-penicillin therapy irrespective of penicillin allergy. Further work is necessary to determine the reason for the increase in return visits.
Funding: None
Disclosures: None
Update on Improving Outpatient Antibiotic Use Through Implementation and Evaluation of Core Elements of Outpatient Antibiotic
- Karl Madaras-Kelly, Christopher Hostler, Mary Townsend, Emily Potter, Emily Spivak, Sarah Hall, Matthew Goetz, McKenna Nevers, Jian Ying, Benjamin Haaland, Suzette Rovelsky, Benjamin Pontefract, Katherine Fleming-Dutra, Matthew Samore
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s422
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Acute respiratory infections (ARIs) are a key target to improve antibiotic use in the outpatient setting. The Core Elements of Outpatient Antibiotic Stewardship provide a framework for improving antibiotic use, but data on safety and effectiveness of interventions to improve antibiotic use are limited. We report the impact of Core Elements implementation within Veterans’ Healthcare Administration clinics on antibiotic prescribing and patient outcomes. Methods: The intervention targeting treatment of uncomplicated ARIs (sinusitis, pharyngitis, bronchitis, and viral upper respiratory infections [URIs]) in emergency department and primary care settings was initiated within 10 sites between September 2017 and January 2018. The intervention was developed using the Core Elements and included local site champions, audit-and-feedback with peer comparison, and academic detailing. We evaluated the following outcomes: per-visit antibiotic prescribing rates overall and by diagnosis; appropriateness of treatment; 30-day ARI revisits; 30-day infectious complications (eg,, pneumonia,); 30-day adverse medication effects; 90-day Clostridium difficile infection (CDI); and 30-day hospitalizations. Multilevel logistic regression was used to calculate rate ratios (RR) with 95% CI for each outcome in the postintervention period (12 months) compared to the preintervention period (39–42 months). Results: There were 14,020 uncomplicated ARI visits before the intervention and 4,866 uncomplicated ARI visits after the intervention. The proportions of uncomplicated ARI visits with antibiotics prescribed were 59.17% before the intervention versus 44.34% after the intervention. A trend in reduced antibiotic prescribing for ARIs throughout the entire (before and after) observation period was evident (0.92; 95% CI, 0.90–0.94); however, a significant reduction in antibiotic prescribing after the intervention was identified (0.74; 95% CI, 0.59–0.93). Per-visit antibiotic prescribing rates decreased significantly for bronchitis and URI (0.54; 95% CI, 0.44–0.65), pharyngitis (0.76; 95% CI, 0.67–0.86), and sinusitis (0.92; 95% CI, 0.85–1.0). Appropriate therapy for pharyngitis increased (1.43; 95% CI, 1.21–1.68), but appropriate therapy for sinusitis remained unchanged (0.92; 95% CI, 0.85–1.0) after the intervention. Complications associated with antibiotic undertreatment were not different after the intervention: ARI-related revisit rates (1.01; 95% CI, 0.98–1.05) and infectious complications (1.01; 95% CI, 0.79–1.28). A potential benefit of improved antibiotic use included a reduction in visits for adverse medication effects (0.82; 95% CI, 0.72–0.94). Furthermore, 90-day CDI events were too sparse to model: preintervention incidence was 0.08% and postintervention incidence was 0.06%. Additionally, 30-day hospitalizations were significantly lower in the postintervention period (0.79; 95% CI, 0.72–0.87). Conclusions: Implementation of the Core Elements was safe and effective and was associated with reduced antibiotic prescribing rates for uncomplicated ARIs, improvements in diagnosis-specific appropriate therapy, visits for adverse antibiotic effects, and 30-day hospitalization rates. No adverse events were noted in ARI-related revisit rates or infectious complications. CDI rates were low and unchanged.
Funding: None
Disclosures: None
VA Antibiotic Stewardship Intervention to Improve Outpatient Antibiotic Use for ARIs: A Cost-Effectiveness Analysis
- Minkyoung Yoo, Richard Nelson, McKenna Nevers, Karl Madaras-Kelly, Katherine Fleming-Dutra, Adam Hersh, Jian Ying, Benjamin Haaland, Matthew Samore
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s55
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: The Core Elements of Outpatient Antibiotic Stewardship provide a framework to improve antibiotic use, but cost-effectiveness data on interventions to improve antibiotic use are limited. Beginning in September 2017, an antibiotic stewardship intervention was launched in within 10 outpatient Veterans Healthcare Administration clinics. The intervention was based on the Core Elements and used an academic detailing (AD) and an audit and feedback (AF) approach to encourage appropriate use of antibiotics. The objective of this analysis was to evaluate the cost-effectiveness of the intervention among patients with uncomplicated acute respiratory tract infections (ARI). Methods: We developed an economic simulation model from the VA’s perspective for patients presenting for an index outpatient clinic visit with an ARI (Fig. 1). Effectiveness was measured as quality-adjusted life-years (QALYs). Cost and utility parameters for antibiotic treatment, adverse drug reactions (ADRs), and healthcare utilization were obtained from the published literature. Probability parameters for antibiotic treatment, appropriateness of treatment, antibiotic ADRs, hospitalization, and return ARI visits were estimated using VA Corporate Data Warehouse data from a total of 22,137 patients in the 10 clinics during 2014–2019 before and after the intervention. Detailed cost data on the development of the AD and AF materials and electronically captured time and effort for the National AD Service activities by specific providers from a national ARI campaign were used as a proxy for the cost estimate of similar activities conducted in this intervention. We performed 1-way and probabilistic sensitivity analyses (PSAs) using 10,000 second-order Monte Carlo simulations on costs and utility values using their means and standard deviations. Results: The proportion of uncomplicated ARI visits with antibiotics prescribed (59% vs 40%) was lower and appropriate treatment was higher (24% vs 32%) after the intervention. The intervention was estimated to cost $110,846 (2018 USD) over a 2-year period. Compared to no intervention, the intervention had lower mean costs ($880 vs $517) and higher mean QALYs (0.837 vs 0.863) per patient because of reduced inappropriate treatment, ADRs, and subsequent healthcare utilization, including hospitalization. In threshold analyses, the antibiotic stewardship strategy was no longer dominant if intervention cost was >$64,415,000 or the number of patients cared for was <3,672. In the PSA, the antibiotic stewardship intervention was dominant in 100% of the 10,000 Monte Carlo iterations (Fig. 2). Conclusions: In every scenario, the VA outpatient AD and AF antibiotic stewardship intervention was a dominant strategy compared to no intervention.
Funding: None
Disclosures: None
Development of an Electronic Tool to Measure Daily Appropriateness of Inpatient Antibacterial Use
- Vanessa Stevens, Pamela Belperio, Melinda Neuhauser, Lauri Hicks, McKenna Nevers, Matthew Goetz, Nai-Chung Chang, Arjun Srinivasan, Makoto Jones
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s2
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Assessing antimicrobial use (AU) appropriateness isa cornerstone of antimicrobial stewardship, largely accomplished through time-intensive manual chart review of specific agents or diagnoses. Efforts to evaluate appropriateness have focused on assessing the appropriateness of an entire treatment course. An electronic measure was developed to assess the appropriateness of each day of inpatient AU leveraging electronic health record data. Methods: We extracted contextual data, including risk factors for resistant organisms, allergies, constitutional signs and symptoms from diagnostic and procedural codes, and microbiological findings, from the electronic health records of patients in Veterans’ Health Administration inpatient wards reporting data to the National Healthcare Safety Network (NHSN) AU option from 2017–2018. Only the antibacterial categories shown in Figure 1 were included. Respiratory, urinary tract, skin and soft-tissue, and other infection categories were defined and applied to each hospital day. Algorithm rules were constructed to evaluate AU based on the clinical context (eg, in the ICU, during empiric therapy, drug–pathogen match, recommended drugs, and duration). Rules were drawn from available literature, were discussed with experts, and were then refined empirically. Generally, the rules allowed for use of first-line agents unless risk factors or contraindications were identified. AU was categorized as appropriate, inappropriate, or indeterminate for each day, then aggregated into an overall measure of facility-level AU appropriateness. A validation set of 20 charts were randomly selected for manual review. Results: Facility distribution of appropriateness, inappropriateness, and indeterminate AU by 4 of the adult, 2017 baseline NHSN Standardized Antimicrobial Administration Ratio (SAAR) categories are shown in Figure 1. The median facility-level inappropriateness across all SAAR categories was 37.2% (IQR, 29.4%–52.5%). The median facility-level indeterminate AU across all SAAR categories was 14.4% (IQR, 9.1%–21.2%). Chart review of 20 admissions showed agreement with algorithm appropriateness and inappropriateness in 95.4% of 240 antibacterial days.
Conclusions: We developed a comprehensive, flexible electronic tool to evaluate AU appropriateness for combinations of setting, antibacterial agent, syndrome, or time frame of interest (eg, empiric, definitive, or excess duration). Application of our algorithm in 2 years of VA acute-care data suggest substantial interfacility variability; the highest rates of inappropriateness were for anti-MRSA therapy. Our preliminary chart review demonstrated agreement between electronic and manual review in >95% of antimicrobial days. This approach may be useful to identify potential stewardship targets, in the development of decision support systems, and in conjunction with other metrics to track AU over time.
Funding: None
Disclosures: None
Effectiveness of a multisite personal protective equipment (PPE)–free zone intervention in acute care
- Lindsay D. Visnovsky, Yue Zhang, Molly K. Leecaster, Nasia Safdar, Lauren Barko, Candace Haroldsen, Diane L. Mulvey, McKenna Nevers, Catherine Shaughnessy, Kristina M. Stratford, Frank A. Drews, Matthew H. Samore, Jeanmarie Mayer
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 40 / Issue 7 / July 2019
- Published online by Cambridge University Press:
- 07 June 2019, pp. 761-766
- Print publication:
- July 2019
-
- Article
- Export citation
-
Objective:
Determine the effectiveness of a personal protective equipment (PPE)-free zone intervention on healthcare personnel (HCP) entry hand hygiene (HH) and PPE donning compliance in rooms of patients in contact precautions.
Design:Quasi-experimental, multicenter intervention, before-and-after study with concurrent controls.
Setting:All patient rooms on contact precautions on 16 units (5 medical-surgical, 6 intensive care, 5 specialty care units) at 3 acute-care facilities (2 academic medical centers, 1 Veterans Affairs hospital). Observations of PPE donning and entry HH compliance by HCP were conducted during both study phases. Surveys of HCP perceptions of the PPE-free zone were distributed in both study phases.
Intervention:A PPE-free zone, where a low-risk area inside door thresholds of contact precautions rooms was demarcated by red tape on the floor. Inside this area, HCP were not required to wear PPE.
Results:We observed 3,970 room entries. HH compliance did not change between study phases among intervention units (relative risk [RR], 0.92; P = .29) and declined in control units (RR, 0.70; P = .005); however, the PPE-free zone did not significantly affect compliance (P = .07). The PPE-free zone effect on HH was significant only for rooms on enteric precautions (P = .008). PPE use was not significantly different before versus after the intervention (P = .15). HCP perceived the zone positively; 65% agreed that it facilitated communication and 66.8% agreed that it permitted checking on patients more frequently.
Conclusions:HCP viewed the PPE-free zone favorably and it did not adversely affect PPE or HH compliance. Future infection prevention interventions should consider the complex sociotechnical system factors influencing behavior change.









